Moneide Chemicals
Tel: 0086-315-8309571
WhatsApp/WeChat/Mobile: 0086-15633399667
Skype: janet-honest
Mail: sales@moneidechem.com
Address: 2-7-523 Jidong Building Materials Commercial Center, Tangshan, Hebei 064000 China
Salicylaldoxime Structure Explained – Applications & Industry Insights
- Time of issue:Nov . 29, 2025 03:50
(Summary description)Tangshan Moneide Trading Co., Ltd. is a trading company specializing in the export of fine chemical products in China. Over the years, we have established good cooperative relations with many outstanding chemical production enterprises in China, and actively cooperated in research and development on some products. Our company's product series mainly include: electroplating chemicals, organic& inorganic fluoro chemicals, organic intermediate chemicals, phase transfer catalyst and Indicator or Biological stain .
- Categories:Company dynamic
- Author:
- Origin:
- Time of issue:2019-12-30 10:55
- Views:
If you’ve ever glanced through a chemistry manual or dived into metal extraction processes, chances are you’ve bumped into the term salicylaldoxime structure. Though it might sound like a mouthful, this chemical structure plays an outsized role in industries ranging from mining to environmental remediation. Globally, industries seek efficiency paired with environmental consciousness: here, the salicylaldoxime structure offers a gentle handshake between performance and sustainability. Grasping what it is and why it matters can unlock smarter applications and innovation – but more on that soon.
The mining industry alone accounts for nearly 18 million jobs worldwide and significantly impacts GDP, as reported by the United Nations Environment Programme (UNEP). The need to extract metals like copper and nickel efficiently, with minimal environmental fallout, is increasingly urgent. Salicylaldoxime, part of the oxime family, acts as a key agent in solvent extraction methods, especially in hydrometallurgy — a practice critical to purifying metals for electronics, renewable energy, and infrastructure.
Yet, the challenge is steep: conventional extraction can be wasteful or toxic. The nuanced makeup of the salicylaldoxime structure allows for selective bonding with metals, minimizing chemical waste and energy consumption. With global metal demand projected to rise by around 6% annually (World Bank data), such chemistry isn’t just useful — it’s essential.
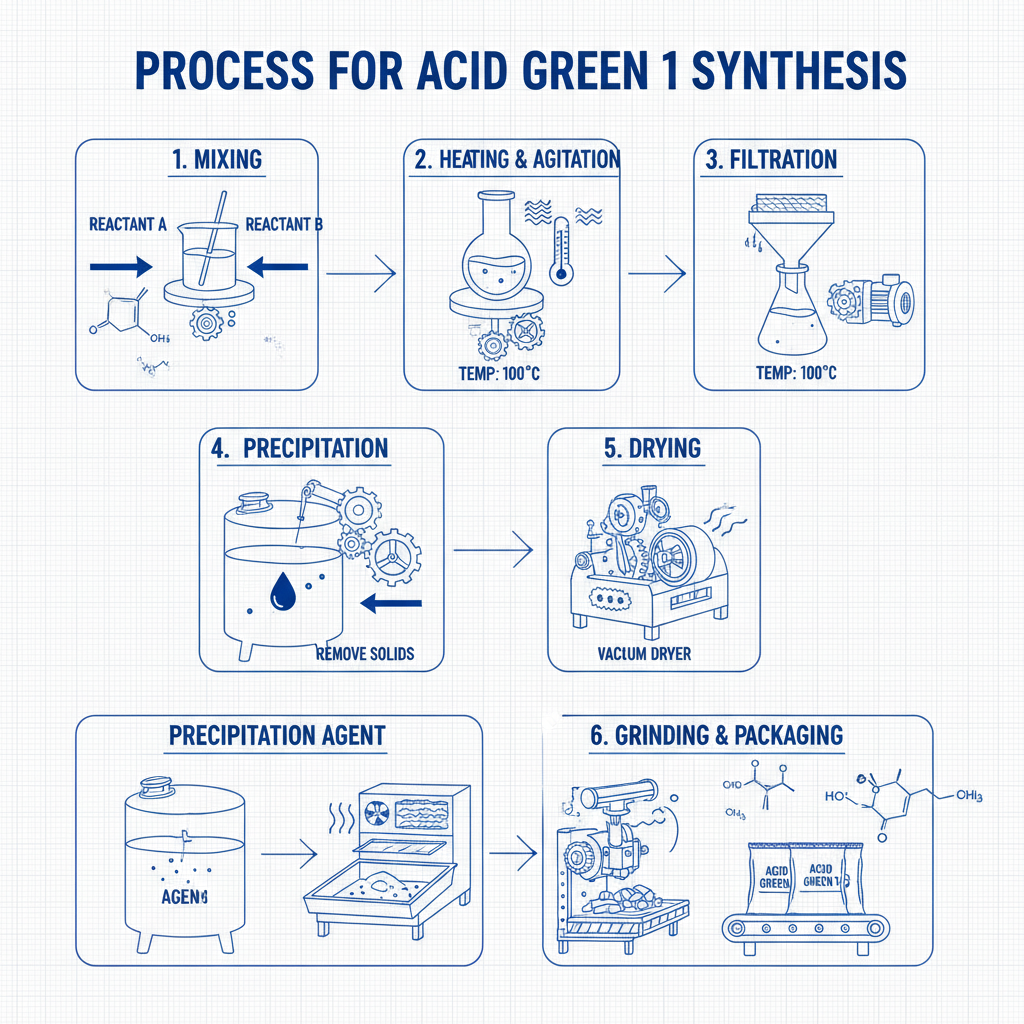
At its core, salicylaldoxime structure refers to a chemical compound derived from salicylaldehyde and hydroxylamine. Picture a molecule with a benzene ring connected to an oxime group (-C=NOH) and a hydroxyl (-OH) attached nearby — a nifty arrangement that enables it to latch on selectively to certain metal ions.
This configuration is pivotal for its chelating abilities. In practical terms, it acts sort of like a custom-made claw that grabs specific metal atoms — say, copper or nickel — while ignoring others. This is invaluable in industries where you want to separate valuable metals from unwanted sludges or ores.
You might think this is purely lab talk, but chemicals with this distinctive structure support everything from refining batteries to safeguarding clean water supplies.
The salicylaldoxime structure’s molecular design favors coordination with copper ions, among others. This selectivity makes it an excellent extractant in solvent extraction processes, reducing impurities.
Many extraction agents degrade or lose efficacy when exposed to heat and acidic or basic environments. Salicylaldoxime derivatives, however, demonstrate remarkable chemical stability, ensuring longevity in demanding setups.
Compared to older, more toxic reagents, salicylaldoxime-based agents typically exhibit lower toxicity and produce less hazardous waste, aligning with ISO standards on sustainable chemical use.
Because of its efficiency and reusability, the salicylaldoxime structure reduces material consumption and downstream processing, ultimately cutting operational costs for companies.
In a nutshell, the salicylaldoxime structure’s beauty lies in its precision — a molecular tightrope walker that balances performance with eco-friendliness.
Around the world, salicylaldoxime compounds underpin many hydrometallurgical processes. In Chile and Peru, copper mining giants leverage these agents to streamline copper recovery, boosting yield and reducing chemical spillage. In Asia, salicylaldoxime finds its role in recycling operations — reclaiming nickel from electric vehicle batteries, for instance.
Industrial zones in Europe use these chemicals to refine copper cathodes with enhanced purity, directly benefiting renewable energy manufacturers who need reliable, high-quality metals. Even humanitarian organizations eye the potential, using simplified extraction kits with salicylaldoxime analogs to recover metals in resource-poor settings, where traditional mining infrastructure is lacking.
From sprawling mines to small-scale recovery workshops, the salicylaldoxime structure is quietly powering progress worldwide.
Industries adopting salicylaldoxime-based extraction report several advantages they swear by. First, cost savings: because the molecule offers high selectivity, companies save on downstream purification steps, minimizing waste disposal fees. Then, there’s environmental impact. Less toxic residue means less soil and water contamination — a real boon in sensitive ecosystems.
On a human level, that means better safety for workers and local communities, plus the comfort of knowing technology is moving towards responsible stewardship. Many engineers I talked to admit it feels like chemistry is finally getting ahead of nature's curve instead of lagging behind.
What’s next? Research is buzzing around tailoring the salicylaldoxime structure for even more efficient recovery of critical metals used in green tech — think cobalt and lithium for batteries. Advances in nanotechnology are leading to nano-scale salicylaldoxime derivatives that dramatically increase surface area and binding.
Artificial intelligence is also helping chemists model new compounds faster than ever before, speeding innovation cycles. Industry standards increasingly encourage green chemistry principles, pushing the spotlight firmly onto agents like salicylaldoxime. It’s an exciting time, where chemistry isn’t just about reactions—it’s about ecosystems, economies, and ethics.
Not everything is smooth sailing. Handling salicylaldoxime chemicals requires precise process control to maintain their stability and avoid degradation. In some regions, supply chain delays can stall operations, especially where purity grade is critical.
To counter these hurdles, vendors have begun offering stabilized formulations and consulting services for process optimization. Recycling solvent phases and reusing salicylaldoxime extracts inside closed-loop systems is becoming industry best practice — a small complexity upfront that reaps huge sustainability rewards long-term.
The molecular configuration allows it to selectively form strong complexes with copper and nickel ions, enabling efficient separation from ore leachates without affecting other metals. This selective binding reduces impurities and waste.
Salicylaldoxime and its derivatives maintain chemical stability in a range of temperatures and acidic conditions commonly found in solvent extraction plants, ensuring consistent performance throughout the process.
Yes, modern hydrometallurgical systems incorporate solvent recycling that allows salicylaldoxime compounds to be recovered and reused multiple times, significantly reducing chemical consumption and environmental impact.
Generally, salicylaldoxime-based reagents are less toxic than older extraction agents, but safe handling and disposal practices are essential. Many manufacturers comply with ISO and REACH regulations to ensure environmental standards.
Compare purity, compliance certifications, lead times, and post-sales support. Vendors specializing in customized formulations or technical consulting often provide better long-term value.
Salicylaldoxime structure is more than an academic term — it’s a cornerstone of modern extraction chemistry that blends selectivity, sustainability, and cost-efficiency. Its flexibility in applications from copper mining to battery recycling makes it a quiet hero behind many technological advances.
If your industry deals with metals or is looking for greener chemistry options, diving deeper into the salicylaldoxime structure and its derivatives could revolutionize your processes — or at least make you appreciate the elegance of molecular design a lot more.
For more insights and sourcing info, be sure to visit our page on salicylaldoxime structure.
Understanding the Salicylaldoxime Structure: Why it Matters in Modern Chemistry
A Global Perspective: The Rising Importance of Salicylaldoxime
What Is the Salicylaldoxime Structure? A Simple Definition
Key Features of the Salicylaldoxime Structure
1. Selective Metal Binding
2. Stability Under Industrial Conditions
3. Environmental Compatibility
4. Cost-Effectiveness
Mini Takeaway:
Global Applications & Real-World Use Cases
Mini Takeaway:
Salicylaldoxime Structure: Product Specification Table
Property
Details
Molecular Formula
C₇H₇NO₂
Molecular Weight
137.14 g/mol
Functional Groups
Hydroxyl (-OH), Aldoxime (-C=NOH)
Solubility
Slightly soluble in water, soluble in organic solvents
Melting Point
107-109 °C
Industrial Use
Copper and nickel solvent extraction
Comparing Leading Vendors of Salicylaldoxime-Based Agents
Vendor
Purity (%)
Bulk Price (per kg)
Environmental Certification
Typical Lead Time
ChemPure Ltd.
99.5
$45
ISO 14001
2 weeks
GreenChem Solutions
98.8
$50
REACH Compliant
3 weeks
Metal Extract Corp.
99.9
$48
ISO 9001 & 14001
1 week
Advantages & Long-Term Value
Emerging Trends & Innovations
Challenges & Solutions in Practical Use
FAQ: Frequently Asked Questions About Salicylaldoxime Structure
Q1: What makes the salicylaldoxime structure effective in metal extraction?
Q2: How stable is salicylaldoxime during industrial processing?
Q3: Can salicylaldoxime-based agents be recycled or reused?
Q4: Are there environmental concerns associated with salicylaldoxime?
Q5: How do I choose the right supplier for salicylaldoxime?
Conclusion: Why the Salicylaldoxime Structure Deserves Your Attention
References