Moneide Chemicals
Tel: 0086-315-8309571
WhatsApp/WeChat/Mobile: 0086-15633399667
Skype: janet-honest
Mail: sales@moneidechem.com
Address: 2-7-523 Jidong Building Materials Commercial Center, Tangshan, Hebei 064000 China
High-Purity Drug Intermediates | Bulk & Custom Solutions
- Time of issue:Sep . 22, 2025 12:10
(Summary description)Tangshan Moneide Trading Co., Ltd. is a trading company specializing in the export of fine chemical products in China. Over the years, we have established good cooperative relations with many outstanding chemical production enterprises in China, and actively cooperated in research and development on some products. Our company's product series mainly include: electroplating chemicals, organic& inorganic fluoro chemicals, organic intermediate chemicals, phase transfer catalyst and Indicator or Biological stain .
- Categories:Company dynamic
- Author:
- Origin:
- Time of issue:2019-12-30 10:55
- Views:
The pharmaceutical industry relies heavily on a complex ecosystem of chemical compounds to develop life-saving medications. At the heart of this process are drug intermediates, specialized chemical substances that are crucial building blocks for active pharmaceutical ingredients (APIs). These intermediates are not the final drug product but are essential compounds formed during the synthesis of an API from its initial raw materials. Their quality, purity, and consistency directly impact the safety, efficacy, and manufacturability of the final drug substance. Our focus herein is to elucidate the critical role, technical specifications, and strategic considerations surrounding these pivotal compounds.
The demand for high-quality drug intermediates is escalating due to continuous advancements in pharmaceutical research and development, alongside increasingly stringent regulatory requirements globally. As pharmaceutical companies strive for greater efficiency, cost-effectiveness, and compliance, the selection and procurement of reliable intermediate suppliers become paramount. This comprehensive overview delves into the intricate world of drug intermediates, providing B2B decision-makers and technical engineers with insights into industry trends, technical advantages, customization capabilities, and real-world application scenarios.
The market for bulk drug intermediates is characterized by several dynamic trends. Global pharmaceutical manufacturing is witnessing a shift towards specialized and high-potency APIs, which in turn drives the need for more sophisticated and precisely synthesized intermediates. Key trends include:
These trends highlight the critical need for suppliers of bulk drug intermediates to be agile, innovative, and deeply committed to quality and regulatory adherence.
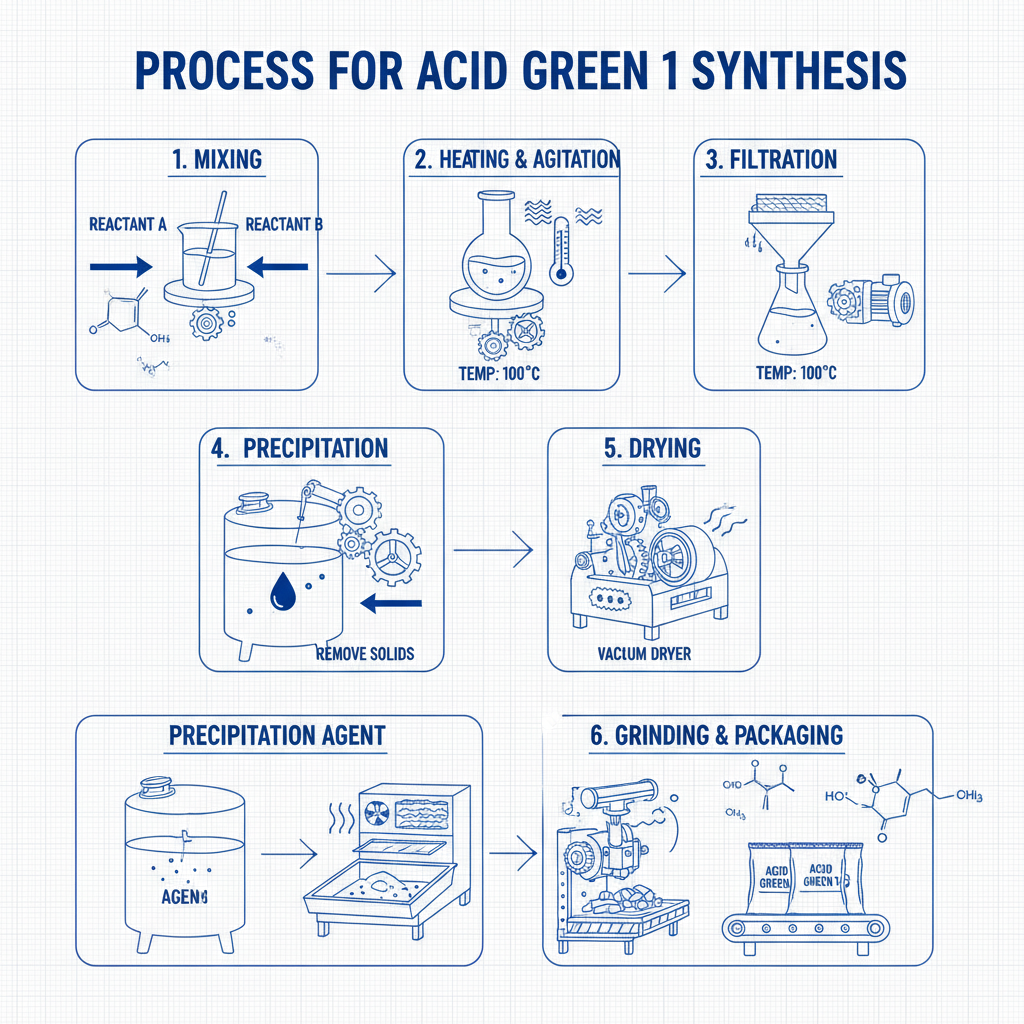
The synthesis of drug intermediates is a multi-step chemical process requiring meticulous control at each stage to ensure the desired purity, yield, and stereoselectivity. While specific processes vary greatly depending on the intermediate's chemical structure, a generalized flow typically involves:
Target industries for these advanced chemical intermediates span a wide range, primarily focused on pharmaceutical manufacturing, but also extending to specialized chemical synthesis for agrochemicals and advanced materials. The typical service life of an intermediate refers more to its stability under specified storage conditions, which can range from 2 to 5 years, validated through rigorous stability studies.
The performance and suitability of drug intermediates are defined by a stringent set of technical specifications. These parameters are critical for ensuring the intermediate will perform as expected in subsequent synthesis steps and contribute to a high-quality API.
These detailed specifications, validated through stringent testing standards (e.g., ISO/IEC 17025 accredited labs, internal GMP-compliant QC), ensure that each batch of drug intermediates meets the highest industry benchmarks for quality and consistency.
Drug intermediates are foundational in a myriad of pharmaceutical applications. Their technical advantages are often tied to their purity, defined stereochemistry, and reactive functionalities, enabling efficient and selective synthesis of complex APIs.
For instance, the precise control over the synthesis of an intermediate like (S)-N-(2,6-Dimethylphenyl)-2-piperidinecarboxamide provides a distinct advantage in producing highly enantiomerically pure APIs, which are often the standard in modern pharmaceutical development. This precision translates directly into energy saving due to fewer purification cycles and reduced solvent usage.
Choosing the right supplier for bulk drug intermediates is a strategic decision that impacts product quality, supply chain reliability, and regulatory compliance. Key differentiators beyond price include technical expertise, quality assurance, and scalability.
MoneideChem stands out by combining robust quality control with advanced synthetic capabilities, offering superior purity and technical support, especially for complex chiral drug intermediates. Our long-standing commitment to quality and comprehensive regulatory documentation minimizes risks for our partners.
The dynamic nature of pharmaceutical R&D often necessitates unique chemical structures or modifications to existing drug intermediates. Custom synthesis services are invaluable in this context.
Our approach to customized solutions for bulk drug intermediates involves:
This bespoke service model allows pharmaceutical companies to access precisely tailored drug intermediates, accelerating their R&D efforts and streamlining their API manufacturing processes, whether for early-stage discovery or late-stage commercialization.
Our commitment to delivering high-quality drug intermediates is validated through successful partnerships and positive customer feedback.
A leading oncology pharmaceutical company required a highly complex, enantiomerically pure intermediate for their novel anti-cancer agent, facing challenges with consistency and scalability from their previous supplier. We developed a proprietary, multi-step synthesis route for a custom drug intermediate, achieving >99.8% purity and >99.7% EE consistently across large-scale batches. This partnership enabled the client to significantly reduce their API manufacturing lead time by 25% and achieve superior product yield, ultimately accelerating their drug's journey through clinical trials. "MoneideChem's technical expertise and reliable supply chain were instrumental in overcoming our critical manufacturing bottlenecks," remarked their Head of Process Chemistry.
A major generic pharmaceutical manufacturer sought to optimize the production of a widely used cardiovascular API. Their existing supplier of drug intermediates often provided batches with variable impurity profiles, leading to extensive downstream purification and increased costs. Our team collaborated with them to supply a specific bulk drug intermediate with an ultra-low impurity profile (genotoxic impurities below detection limits). This resulted in an immediate 15% reduction in API purification costs and a 10% increase in overall API yield, demonstrating significant energy saving and improved process robustness.
Q: What are drug intermediates and why are they important?
A: Drug intermediates are chemical compounds formed as part of the synthetic pathway from initial raw materials to the final Active Pharmaceutical Ingredient (API). They are crucial because their quality directly influences the purity, efficacy, and safety of the final drug product, acting as essential building blocks that enable complex API synthesis.
Q: How do you ensure the quality and purity of bulk drug intermediates?
A: We adhere to stringent quality management systems, including ISO 9001:2015 and cGMP guidelines. Our process involves rigorous raw material qualification, in-process controls at every synthesis stage, and comprehensive final product testing using advanced analytical techniques (HPLC, GC, NMR, MS, ICP-MS) to ensure purity, identification, and absence of impurities and heavy metals.
Q: Can MoneideChem provide customized drug intermediates?
A: Yes, we specialize in custom synthesis. Our R&D team works closely with clients to develop and optimize synthetic routes for novel or specific drug intermediates, scale up production from gram to ton quantities, and provide full analytical and regulatory support.
Q: What is the typical lead time for bulk orders?
A: Lead times vary based on the complexity of the intermediate, order quantity, and current production schedules. For standard products, it typically ranges from 4-8 weeks. For custom synthesis or very large bulk orders, lead times will be provided after detailed project assessment, usually between 8-16 weeks for initial production batches. We prioritize efficient fulfillment and transparent communication throughout the process.
MoneideChem stands behind the quality of its drug intermediates with a robust warranty and comprehensive after-sales support.
Our commitment extends beyond delivery, fostering long-term partnerships built on trust and mutual success. For any inquiries, please contact our support team at info@moneidechem.com or visit our website for further details.
Understanding Drug Intermediates in Pharmaceutical Synthesis
Industry Trends and Market Dynamics for Bulk Drug Intermediates
Manufacturing Process Flow of Drug Intermediates
Schematic Steps of Intermediate Synthesis:
Technical Specifications and Parameters for Drug Intermediates

Key Product Specifications (Example: Chiral Intermediate)
Parameter
Specification
Test Method
Product Name
(S)-N-(2,6-Dimethylphenyl)-2-piperidinecarboxamide
Internal Standard
Chemical Formula
C14H20N2O
Calculated
Molecular Weight
232.32 g/mol
Calculated
Appearance
White to off-white crystalline powder
Visual
Purity (HPLC)
≥ 99.0%
HPLC (USP Method)
Enantiomeric Excess (EE)
≥ 99.5%
Chiral HPLC
Water Content
≤ 0.5%
Karl Fischer
Residual Solvents
Conforms to ICH Q3C limits
GC-HS
Heavy Metals
≤ 10 ppm
ICP-MS
Storage Condition
2-8°C, tightly sealed, protect from light
Internal Standard
Application Scenarios and Technical Advantages
Typical Application Scenarios:
Technical Advantages:
Vendor Comparison: Selecting the Right Partner for Drug Intermediates
Comparison Criteria for Drug Intermediate Suppliers:
Feature/Criterion
MoneideChem's Offering
Typical Competitor A (Generic)
Typical Competitor B (Specialized)
Quality Certifications
ISO 9001:2015, cGMP Compliant, ICH Q7 guidelines
ISO 9001:2008 (often), limited GMP adherence
ISO 9001:2015, select GMP compliance, often smaller scale
Purity & Impurity Control
≥ 99.0% purity, comprehensive impurity profiling, low heavy metals
Typically 95-98% purity, basic impurity checks
≥ 98.5% purity, good impurity control for specific products
Chiral Synthesis Capability
Expertise in complex chiral intermediates, high EE (e.g., >99.5%)
Limited chiral capability, lower EE for complex structures
Specialized in certain chiral families, varying EE
Technical Support & R&D
Dedicated R&D team, synthesis route development, process optimization support
Minimal R&D, off-the-shelf products
Moderate R&D, focused on proprietary methods
Scalability (Gram to Ton)
Seamless scale-up from lab to commercial quantities
Often limited to small to mid-scale production
Good for specific products, but broader portfolio scale-up may vary
Documentation & Regulatory Support
Full CoA, MSDS, DMF support (where applicable), audit readiness
Basic CoA, limited regulatory assistance
Good documentation, often for specific regulatory regions
Customized Solutions for Drug Intermediates
Application Case Studies and Customer Experience
Case Study 1: Accelerating Oncology Drug Development
Case Study 2: Optimizing Cardiovascular API Production
Frequently Asked Questions (FAQ) about Drug Intermediates
Warranty and After-Sales Support
Authoritative References