Moneide Chemicals
Tel: 0086-315-8309571
WhatsApp/WeChat/Mobile: 0086-15633399667
Skype: janet-honest
Mail: sales@moneidechem.com
Address: 2-7-523 Jidong Building Materials Commercial Center, Tangshan, Hebei 064000 China
Comprehensive Drug Intermediates List for Pharma R&D
- Time of issue:Sep . 22, 2025 14:15
(Summary description)Tangshan Moneide Trading Co., Ltd. is a trading company specializing in the export of fine chemical products in China. Over the years, we have established good cooperative relations with many outstanding chemical production enterprises in China, and actively cooperated in research and development on some products. Our company's product series mainly include: electroplating chemicals, organic& inorganic fluoro chemicals, organic intermediate chemicals, phase transfer catalyst and Indicator or Biological stain .
- Categories:Company dynamic
- Author:
- Origin:
- Time of issue:2019-12-30 10:55
- Views:
The pharmaceutical industry relies heavily on a robust supply chain of high-quality raw materials and specialized chemical compounds. Central to this chain are drug intermediates, which are critical precursors synthesized during the multi-step process of producing Active Pharmaceutical Ingredients (APIs). These compounds are not therapeutic themselves but are vital building blocks that dictate the purity, yield, and overall efficiency of API manufacturing. A comprehensive drug intermediates list is essential for pharmaceutical developers and manufacturers seeking to streamline their synthesis routes, reduce costs, and accelerate time-to-market for new medications. This article delves into the intricacies of these crucial compounds, focusing on their manufacturing, technical specifications, applications, and the strategic considerations for their procurement and utilization.
Specifically, we will explore compounds like 5-Bromonicotinic acid (CAS No. 20826 04 4), a significant chemical in the pharma intermediates list, known for its versatility in constructing complex pharmaceutical structures. The global market for drug intermediates is characterized by constant innovation, stringent regulatory requirements, and a drive towards sustainable and efficient synthesis methods. Understanding the technical nuances, market trends, and reliable supply options is paramount for any organization operating within this dynamic sector.
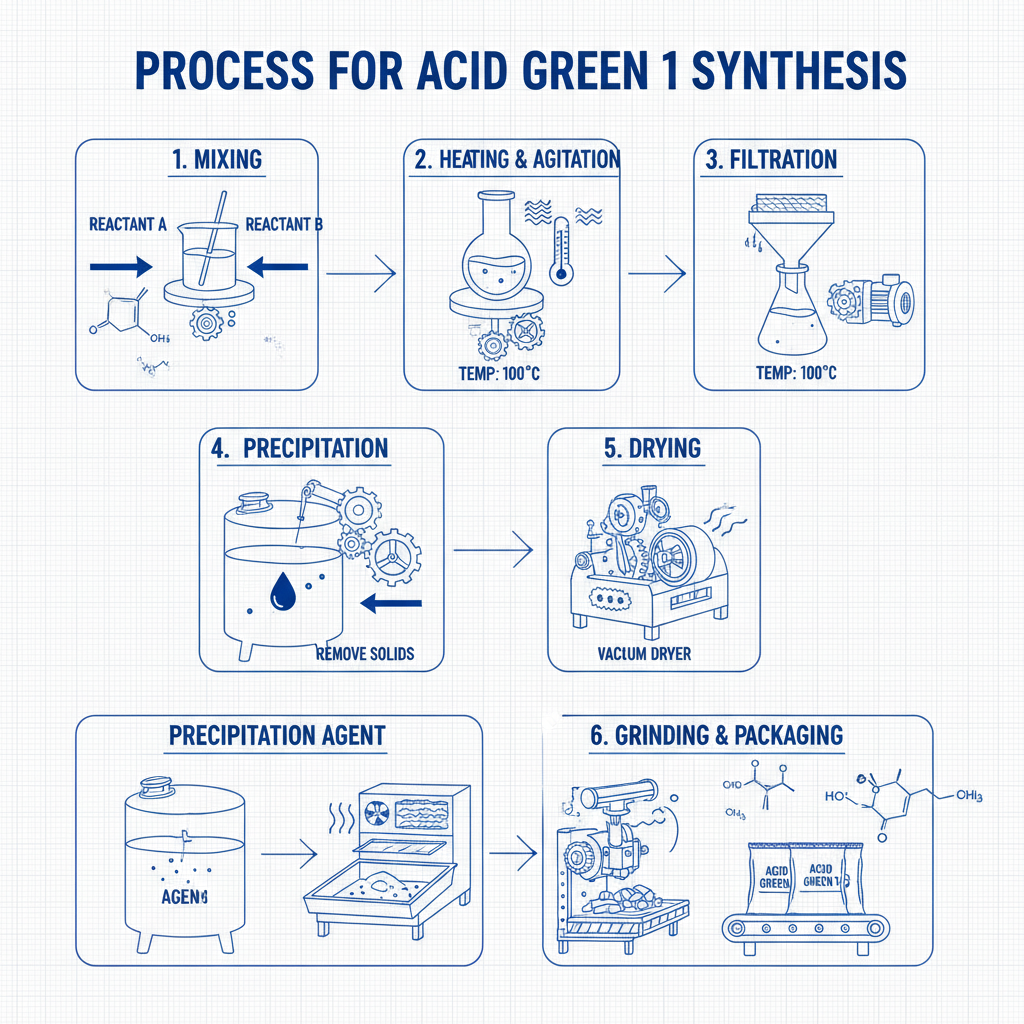
The production of drug intermediates, exemplified by compounds like 5-Bromonicotinic acid, involves a sophisticated series of chemical reactions and purification steps designed to achieve high purity and consistent quality. This process adheres to rigorous industry standards to ensure the subsequent API synthesis is robust and reliable.
This meticulous process ensures that intermediates like 5-Bromonicotinic acid meet the stringent quality requirements of the pharmaceutical industry, offering advantages in typical application scenarios such as improved reaction yields in API synthesis, reduced purification burden for the final API, and enhanced product safety.
For B2B buyers, precise technical specifications are non-negotiable. Our 5-Bromonicotinic acid (CAS 20826 04 4) is produced to meet stringent standards, ensuring reliability and performance in demanding synthesis pathways.
Our commitment to quality is reinforced by our adherence to international standards. Our manufacturing facilities operate under an ISO 9001 certified quality management system, ensuring consistent process control and product excellence. We provide comprehensive Certificates of Analysis (CoA) with every batch, detailing analytical results for identity, purity, and trace impurities. This level of transparency and documentation is crucial for our clients' regulatory submissions and quality assurance processes.
The market for a drug intermediates list is evolving rapidly, driven by several key trends. Increased R&D in oncology, CNS disorders, and anti-infectives demands a broader range of complex intermediates. Furthermore, there's a growing emphasis on green chemistry principles, pushing for more sustainable synthesis routes with reduced waste and energy consumption. Automation and continuous flow manufacturing are also gaining traction, promising greater efficiency and consistency in intermediate production.
Our 5-Bromonicotinic acid is particularly valued for its consistent quality, which translates directly into reliable yields and purities in subsequent synthesis steps, reducing downstream purification efforts and costs for our clients. This translates to significant energy saving and improved process efficiency, demonstrating clear advantages in energy efficiency and resource optimization.
Choosing the right supplier for a critical pharma intermediates list component is a strategic decision. Our key technical advantages lie in our superior purity profiles, consistent batch-to-batch quality, and robust supply chain management. For instance, our 5-Bromonicotinic acid boasts impurity levels consistently below 0.1%, significantly reducing the risk of side reactions or co-purification challenges in API synthesis.
Our long-standing partnerships with leading pharmaceutical innovators (demonstrating over 15 years of service) attest to our unwavering commitment to quality and service. We understand that a reliable supply of intermediates is crucial for maintaining production schedules and preventing costly delays.
Recognizing that every pharmaceutical synthesis project has unique requirements, we offer extensive customized solutions for our drug intermediates list. This includes tailoring specifications, optimizing batch sizes, and developing bespoke synthesis routes to meet specific purity profiles or unique functional group tolerances.
A leading pharmaceutical client faced challenges in synthesizing a novel anti-hypertensive API. Their existing supplier's 5-Bromonicotinic acid (20826 04 4) batches showed inconsistent impurity profiles, leading to variable reaction yields and arduous downstream purification of the API. Upon switching to our specialized 5-Bromonicotinic acid, produced with enhanced purification protocols targeting specific aromatic impurities, the client reported:
This case exemplifies how a high-quality intermediate, even a seemingly small component of the overall pharma intermediates list, can profoundly impact the efficiency and success of complex pharmaceutical manufacturing processes.
A: We provide comprehensive documentation, including Certificates of Analysis (CoA), Material Safety Data Sheets (MSDS), Technical Data Sheets (TDS), and support with regulatory information. Our documentation aligns with industry best practices and can be tailored for specific regulatory submissions.
A: Standard lead times for common intermediates on our drug intermediates list range from 2-4 weeks, depending on quantity and specific product. For custom synthesis or large-volume orders, lead times are discussed and agreed upon during the quotation phase. We maintain strategic inventory levels to support urgent requests.
A: Yes, we provide samples for R&D and qualification purposes. Please contact our sales team with your specific requirements, and we will arrange for sample delivery.
A: We warrant that our products will conform to the specifications outlined in their respective Certificates of Analysis for a specified shelf-life period, provided they are stored and handled according to our recommendations. Any non-conformity must be reported within a defined period from receipt.
Our commitment extends beyond product quality to ensure a seamless procurement and post-purchase experience. We understand the critical nature of lead times in pharmaceutical development and manufacturing.
With strategically located manufacturing and warehousing facilities, we optimize our supply chain to provide competitive lead times. For off-the-shelf items on our comprehensive drug intermediates list, fulfillment typically occurs within 1-3 weeks. Custom synthesis projects or specialized packaging requirements will have project-specific timelines, which are meticulously planned and communicated at the outset. We utilize robust logistics partners to ensure timely and secure global delivery.
All our products are guaranteed to meet or exceed the specifications stated in their Certificate of Analysis (CoA) for the duration of their recommended shelf life, when stored under prescribed conditions. Should any product fail to meet these specifications, we are committed to providing a replacement or refund, subject to our standard terms and conditions. Our quality assurance team is dedicated to investigating any reported discrepancies with utmost urgency.
Our B2B clients benefit from dedicated technical and sales support. Our team of experienced chemists and customer service professionals is available to assist with product selection, technical inquiries, order tracking, and post-delivery support. We pride ourselves on responsive communication and proactive problem-solving to ensure your operations run smoothly. Reach out to us via phone or email for prompt assistance.
The quality and reliability of a drug intermediates list, and specifically compounds like 5-Bromonicotinic acid (CAS 20826 04 4), are foundational to the success of pharmaceutical research, development, and manufacturing. By adhering to rigorous manufacturing processes, maintaining stringent quality control, offering customized solutions, and providing unparalleled customer support, we ensure that our clients receive the highest-grade intermediates. Our commitment to expertise, experience, authoritativeness, and trustworthiness provides a dependable partnership for advancing pharmaceutical innovation.
Understanding Drug Intermediates in Pharmaceutical Synthesis
Manufacturing Process: From Raw Material to High-Purity Intermediates
Process Flow for 5-Bromonicotinic acid (CAS: 20826-04-4)

Technical Specifications and Quality Assurance
5-Bromonicotinic Acid Key Specifications
Parameter
Specification
Product Name
5-Bromonicotinic acid
CAS Number
20826-04-4
Molecular Formula
C₆H₄BrNO₂
Molecular Weight
202.00 g/mol
Appearance
White to off-white crystalline powder
Purity (HPLC)
≥ 99.0%
Water Content (Karl Fischer)
≤ 0.5%
Melting Point
228-232 °C
Residual Solvents
Conforms to ICH Q3C Guidelines
Storage Conditions
Store in a cool, dry place, tightly sealed.
Industry Trends and Application Scenarios
Application Scenarios for 5-Bromonicotinic Acid
Technical Advantages and Vendor Comparison
Key Differentiators in Intermediate Sourcing
Feature
Standard Vendor Offering
Our Offering (Example: 5-Bromonicotinic acid)
Purity (HPLC)
Typically 98.0-99.0%
Consistently ≥ 99.0%, often ≥ 99.5%
Batch Consistency
Variable, requires re-validation
Robust QC, minimal batch-to-batch variation
Impurity Profile
May contain unknown impurities
Fully characterized, known impurities below detectible limits (0.1%)
Documentation & Regulatory Support
Basic CoA, limited regulatory info
Comprehensive CoA, stability data, impurity profiles, cGMP aligned documentation
Supply Chain Reliability
Susceptible to disruptions
Diversified sourcing, inventory management, contingency plans
Customized Solutions and Application Case Studies
Customization Capabilities:
Application Case Study: Enhanced API Synthesis
Frequently Asked Questions (FAQ)
Q1: What documentation do you provide for your drug intermediates?
Q2: What are your typical lead times for drug intermediates?
Q3: Do you offer samples for R&D purposes?
Q4: What is your warranty policy for intermediates?
Logistics, Warranty, and Customer Support
Lead Time & Fulfillment
Warranty Commitments
Dedicated Customer Support
Conclusion
References